Which Of The Following Compounds Has The Highest Boiling Point?
A compound’s boiling point depends on how many bonds it forms with other molecules and the strength of these bonds.
To determine which compound has the highest boiling point, we must break each bond between the compound and its mixtures so that each molecule can form a new bond with the molecule next to it.
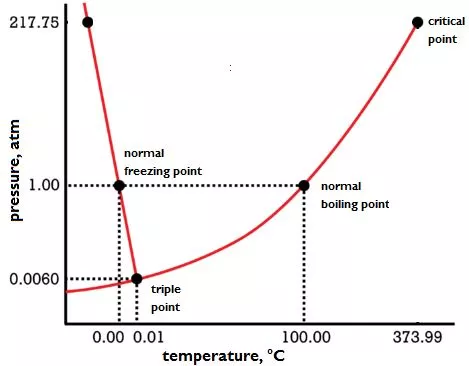
If each compound forms stronger bonds than its neighbors, breaking all of its bonds will take more energy to have the highest boiling point.
Water
This extremely low-density liquid remains a liquid even when heated to more than 100 °C. Water boils when it reaches 100 °C, at which temperature its vapor pressure is high enough to overcome atmospheric pressure.
Even if there is enough heat to boil all of a sample of water, some water will remain in liquid form as a result of phase change latency.
It waits for some external stimulus such as an increase in pressure or decrease in temperature before changing phase (for example, turning from solid ice into liquid water).
Also, note that phase change latency tends to be higher for substances with lower densities and lower heats of vaporization.
For instance, methane’s boiling point is −164 °C, but it still has a higher latent heat of vaporization than water.
Nitrogen can be liquefied by cooling only slightly below its boiling point because of the lack of latent heat required to liquefy it.
It can then be heated as high as 300 °C without boiling because it requires less energy input.
The second is octane, which boils between about 150 °C and 170 °C.
The third would be helium gas, which remains a gas at temperatures below -269 °C (−452 °F), where it becomes a superfluid rather than a vapor.
The fourth would be nitrogen gas; while this inert compound doesn’t have much of a boiling point either (at 77 K), it can become fluid at room temperature under immense pressure.
And finally, the fifth would be methane gas; not only does this compound have the lowest boiling point out of those listed so far (just -164 °C), but due to its larger hydrogen atoms.
Its boiling point falls three times faster than water at standard atmospheric pressure (20 degrees Celsius versus 4 degrees Celsius).
Carbon dioxide
Boiling points are primarily a factor of molecular weight and van der Waals’ forces, so gasses with lighter molecules will have lower boiling points.
Carbon dioxide also forms a powerful intermolecular bond (called a polar covalent bond) because its two bonded atoms each have only one electron to share.
This strong bond between CO2 molecules causes carbon dioxide to have a high melting and boiling point.
With four times as many electrons as carbon dioxide, nitrogen does not form a stable or strong bond with other nitrogen molecules in the atmosphere.
Nitrogen is naturally soluble in water, so it exists mainly as gaseous N2 dissolved in the oceans and lakes.
That is why liquid nitrogen boils at -196 degrees Celsius (-321 degrees Fahrenheit).
The lowest boiling point substances on Earth include hydrogen sulfide (boils at 34 degrees Celsius or 93.2 degrees Fahrenheit), ammonia (boils at -33 degrees Celsius or 26.6 degrees Fahrenheit), and mercury (boils at 357 degrees Celsius or 621.8 degrees Fahrenheit).
Hydrogen chloride boils at 17 C while hydrogen fluoride boils at 24 C. These are highly toxic chemicals you should never handle yourself!
Methane
The boiling point for methane is −162.5 °C, or about 8 degrees Fahrenheit higher than water’s 212 °F.
Light gas is a natural gas product that can be produced at landfills, coal mines, and in cows’ stomachs (believe it or not).
Even though it’s flammable, you may have seen it used as a fuel in aerosol cans, cooking, and fire extinguishers.
Suppose you’re wondering why people don’t just use helium to fill balloons and blimps (or when they need to breathe underwater). In that case, that element boils at -268.94 °C (-452.78 °F), making it useless outside of scientific labs unless you plan on building your cold-resistant container!
With its freezing temperatures, you might think we should replace other fluids with liquid nitrogen in our cars and machinery instead of using fuel.
But unfortunately, liquid nitrogen also contains oxygen molecules, so adding it to an engine would create nitrous oxide, the most potent greenhouse gas after carbon dioxide, rather than reduce pollution.
Adding more methane into our vehicles without dealing with its tricky temperature issues would require innovative solutions.
Perhaps someday, we’ll see mechanical refrigerators working alongside onboard fridges during hot weather days to produce enough vapor pressure to push pistons down while keeping our cars safely at low temperatures.
You might notice that HFC-134A is another example where we’ve made new compounds by mixing gasses like nitrogen and hydrogen with fluorine atoms.
Ammonia
-28.4 °C / -18.0 °F (-34.6 °C / -29.1 °F) at 760 mmHg (compare: Benzene: 40-65°C/ 4-149°F, Ethyl alcohol 64-78°C/ 147-172°F, Glycerol 220-260 C°/ 436-500 F°, Water 100 C°/ 212 F°, Helium 0 K / −273 C°/-458 F°).
There are a whole host of reasons why ammonia is used to preserve things and not just its high boiling point.
One reason is that ammonia does not corrode metals as other solvents do, so that you can store metal objects with it.
It also does not react as quickly with organic materials, so you can store clothes in a container with ammonia for some time without them deteriorating.
People use ammonia to preserve food because it reacts well with salt, which helps inhibit bacterial growth in meat products like bacon or ham.
Final words
Each substance had different atoms, molecules, and bonds that affected its physical properties, such as melting and boiling points.
It is reasonable to assume that based on physical properties, I would expect Sulfur Dioxide to have a lower boiling point than both Carbon Monoxide and Trichlorofluoromethane since it does not contain fluorine and is known for possessing non-polar covalent bonds.



















