Is Cysteine Polar Or Non-Polar?
There are two basic questions that you must answer when you are deciding whether to use a sulfur-containing amino acid in your recipes: is methionine a polar or nonpolar amino acid? Both methionine and cysteine have a sulfur atom in their side chains. The difference between these two amino acids is that methionine has three carbon atoms surrounding it and cysteine has one carbon and one hydrogen atom. When mixing these two amino acids, methionine disrupts the hydrogen bonding between water molecules. This disruption in hydrogen bonding is the difference between a weakly and strongly polar molecule, and very polar molecules can make up for this difference by interacting strongly with water.
Methionine
Methionine and cysteine are two very similar amino acids. However, the polarity of cysteine is much more controversial. Both contain sulfur, a polar atom, in their side chains. Although the polarity of these amino acids differs somewhat, both are highly conserved in proteins. However, the polarity of methionine and cysteine is ambiguous, owing to the fact that their side chains contain three carbon atoms, while cysteine only has one carbon and one hydrogen atom. In fact, the three carbons in methionine are more important than the single hydrogen atom in cysteine, and when the two molecules mix, they disrupt the hydrogen bonding between water molecules. This disruption is due to the fact that weakly polar molecules, such as cysteine and
Methionine and cysteine have distinct roles within proteins. While cysteine is hydrophobic and unable to interact with water, methionine is a nucleophile, and acts as the active site for cysteine proteases, which are typically found near the surface of proteins. In addition, the sulfur in methionine is not highly nucleophilic, but it will react with a number of electrophilic centers. Despite its hydrophobic nature, cysteine does not participate in covalent chemistry of enzyme active centers, resulting in a neutral charge.
Unlike its sulfur cousin, methionine is a polar amino acid. In the human body, it plays several important roles in metabolism, including the detoxification of metabolic wastes. It is one of the few amino acids that is not found in significant amounts in soybeans. Fortunately, many soy meal products add methionine to give it the sulfur it needs to function efficiently.
Lysine
There are two types of amino acids: a molecule with a purely aqueous side chain and a molecule with a polar side chain. A polar amino acid has a side chain with either oxygen or sulfur, which can accept or donate a proton. Other polar amino acids include glutamine, histidine, and lysine. Histidine is a polar amino acid, and it has two -NH groups. Despite its polarity, it is a non-base, and is often classified as a neutral amino acid.
The polar or non-polar cysteine is formed by the oxidation of two cysteines. The thiol group of cysteine is extremely reactive, and one of its most common reactions is oxidation. In fact, cysteine forms a disulfide bond with two Cys-atoms, called a ‘disulfide bridge’. Disulfide bridges stabilize many proteins. They are particularly common in extracellular proteins, where they are highly hydrophobic.
The polar Cys-cysteine hydrophobicity is highly ambiguous. Cysteine is classified as a nonpolar amino acid, and the distinction is based on its presence in the simplest of proteins. Interestingly, Cys-cysteine-containing proteins often have their side chains buried inside the protein. The buried side chains of Cys-containing proteins contain a positively charged side chain, and thus the amino acid prefers to be on the outside.
Another difference between the two types of cysteine is the presence of sulphur-containing amino acids. Cysteine is a hydrophobic amino acid, and is not capable of hydrogen bonds. It interacts with other hydrophobic compounds, the main chain of a protein, and with solvents. It has a much lower affinity to water, but it is hydrophobic.
Threonine
The question of whether Threonine is polar or non-polar arises from its chemical structure. In general, amino acids are polar or non-polar depending on the side chains they have. Some of the more common polar amino acids include serine and arginine. However, there are some exceptions. Histidine is both polar and non-polar depending on its environment. It has two -NH groups and a pKa value of six. It is a polar amino acid because it participates in hydrogen bonding.
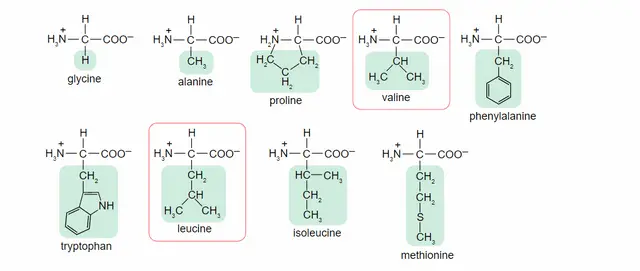
The ‘nonpolar’ class of amino acids contains only those amino acids without hydrogen acceptor or donor atoms. This includes Threonine and other non-polar amino acids such as alanine, valine, leucine, tryptophan, and proline. Other non-polar amino acids include alanine, leucine, valine, and phenylalanine.
As for the ‘nonpolar’ phenylalanine, it is very similar to an aromatic phenylalanine. In fact, it contains an OH group on the para position. However, it has a very small OH group compared to benzyl. Unlike serine, THREEonine has three groups attached: CH, CH3, and OH. Because the side chains are hydrophobic, they are not stable in water and can interact with each other.
Another common amino acid is Glycine. It often occurs in the coil and loop regions of a polypeptide chain. This amino acid provides high flexibility to the polypeptide chain, which is necessary for sharp turns in these regions. Proline, on the other hand, is nonpolar and provides rigidity to the polypeptide chain. A disulfide bridge forms between two cysteine residues and is much stronger than standard tertiary interactions. However, this disulfide bridge can be a source of experimental error when it comes to measuring the size of proteins.
Aspartate
The side chains of amino acids are primarily classified by the polarity of the hydrogen-containing group (R). The hydrophobic Cys group is one of the simplest and least polar, and possesses a hydrogen-based sulfhydryl group on its side chain. Cystine is non-essential for protein synthesis, and is present in beta-keratin, the main protein in the skin and nails.
Cysteine is a semi-essential amino acid with the formula HOOC-CH-CH2-SH. The cysteine thiol side chain often participates in enzymatic reactions, typically as a nucleophile. It also possesses a disulfide derivative, designated with the code name E920, and functions as a structural component of many proteins.
Despite its side-chains containing sulfur and oxygen, cysteine is neither polar nor basic. Proline, for example, originates from glutamic acid. Upon reduction of the carboxy group to an aldehyde, the amino group attacks the corresponding Schiff base. Proline’s N is an alpha-amino group, and tryptophan’s side-chain contains an indole substituent. Because it contains no free lone pairs, tryptophan and tyrosine are classified as nonpolar aromatics.
As mentioned above, the side-chains of amino acids influence their polarity. The more alkyl groups a side chain contains, the less polar it is. For example, serine has more methyl groups than threonine, and it has a higher degree of nonpolarity. The same is true for glycine, which is a non-polar amino acid.
Amino acids have two main types, acidic and basic. Acidic amino acids, such as glycine, have hydrocarbon-containing side chains and are not capable of hydrogen bonding. Non-polar amino acids are the glycine, leucine, and asparagine. The hydroxyl group of glutamine is not polar, and histidine has a charge of +1.
Aspartate disulfide
Whether aspartate disulfide is a cyclic or noncyclic substance depends on the conformation of the amino acid. Cyclic forms of cysteine are polar because they rotate in a levorotary direction, which is opposite to the right-handed rotation. Cyclic forms of aspartate are nonpolar because they do not form covalent disulfide bonds.
Cysteine molecules can form covalent disulfide bonds between two cysteine molecules. These disulfide bonds affect protein stability and folding. In the laboratory, beta-mercaptoethanol is used to break the disulfide bond. The disulfide bonding process is an enzyme-mediated reaction. For the sake of clarity, we will discuss both polar and nonpolar forms of aspartate.